Application of laser marking in pharmaceutical industry
A label is printed on a key component of each medical device. The tag provides a record of where the work was made and can help track it down in the future. Labels usually include the identification of the manufacturer, the production lot and the equipment itself. All medical device manufacturers are required to place permanent and traceable marks on their products for a number of reasons, including product liability and safety.
World medical device regulations require devices and manufacturers to be identified by labels. In addition, labels must be provided in a human-readable format, but they can be supplemented by machine-readable information. Almost all types of medical products must be labeled, including implants, surgical instruments and disposable products, including intubations, catheters and hoses.
CHUKE’s Marking Solutions for Medical and Surgical Instruments
Fiber laser marking is the most suitable technology for defect-free equipment marking. Fiber laser labeled products can be appropriately identified and tracked throughout their life cycle, improving patient safety, simplifying product recalls and improving market research. Laser marking is suitable for identifying marks on medical devices such as orthopaedic implants, medical supplies and other medical devices because the marks are resistant to corrosion and withstand intense sterilization processes, including centrifugation and autoclaving processes that require high temperatures to obtain sterile surfaces.
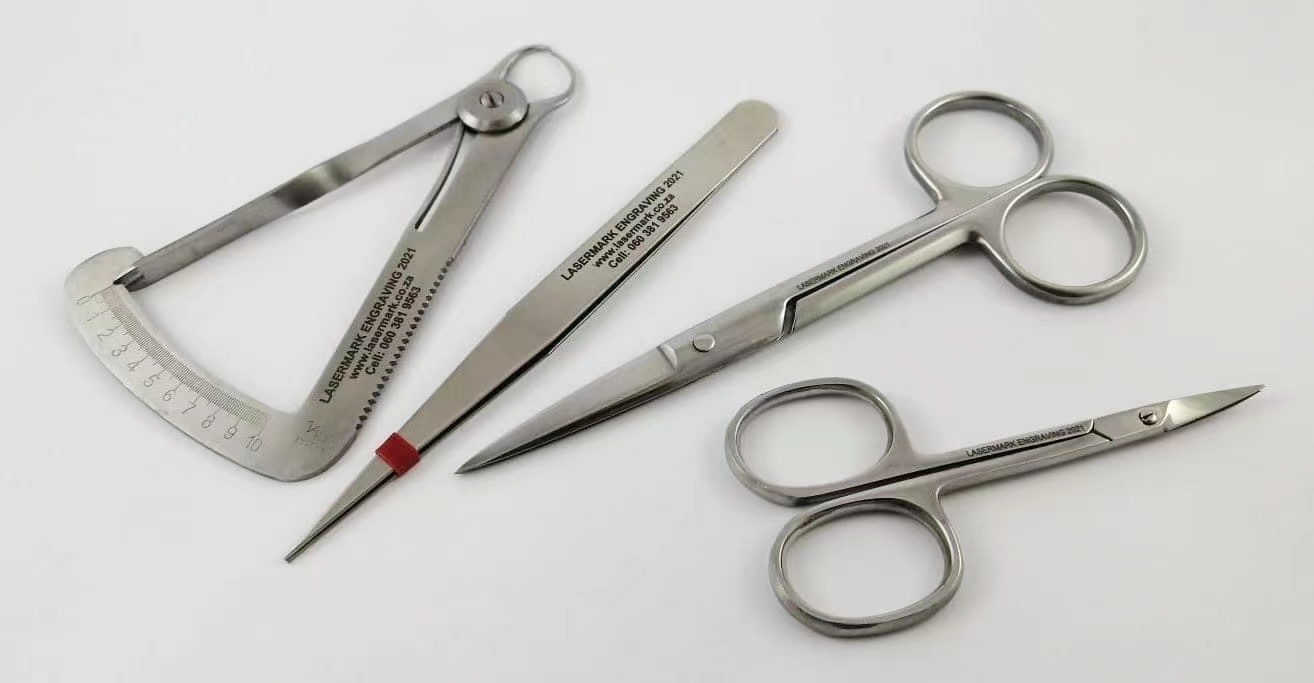



Fiber laser marking is an alternative to etching or engraving treatments, both of which alter the microstructure of the material and can lead to changes in strength and hardness. Because fiber laser marking is non-contact engraving and works quickly, parts do not have to undergo the stress and possible damage that other marking solutions can cause. A dense cohesive oxide coating that "grows" on the surface; You don't need to melt.
Government guidelines for Unique Device Identification (UDI) for all medical devices, implants, tools and devices define permanent, clear and accurate labeling. While tagging improves patient safety by reducing medical errors, providing access to relevant data and facilitating device traceability, it is also used to combat counterfeiting and fraud.
Counterfeiting is a multi-billion dollar market. Fiber laser marking machines provide UDI that distinguishes manufacturer, product era and serial number, which helps combat counterfeit suppliers. Counterfeit equipment and medicines are often sold at much lower prices but of questionable quality. This not only puts patients at risk, but also affects the integrity of the original manufacturer's brand.
CHUKE’s Marking Machine Give You The Best Service
CHUKE fiber optic markers have a small footprint and a service life of between 50,000 and 80,000 hours, so they are very convenient and offer good value to customers. In addition, these laser devices do not use harsh chemicals or high temperatures in the marking process, so they are environmentally sound. This way you can permanently laser mark a variety of surfaces, including metals, stainless steel, ceramics and plastics.